Our focus
Over the last several decades we have studied the structure and function of the myosin family of molecular motors in vitro and in vivo.
That work has led us to our current focus on the human cardiac sarcomere and the molecular basis of hypertrophic and dilated cardiomyopathy.
The myosin mesa hypothesis
We postulated in 2015 that a majority of hypertrophic cardiomyopathy mutations are likely to be shifting beta-cardiac myosin heads from a sequestered off-state to an active on-state for interaction with actin, resulting in the hyper-contractility seen clinically in HCM patients. This hypothesis is different from earlier prevailing views, and this viewing an old disease in a new light is the basis of all of our current research.
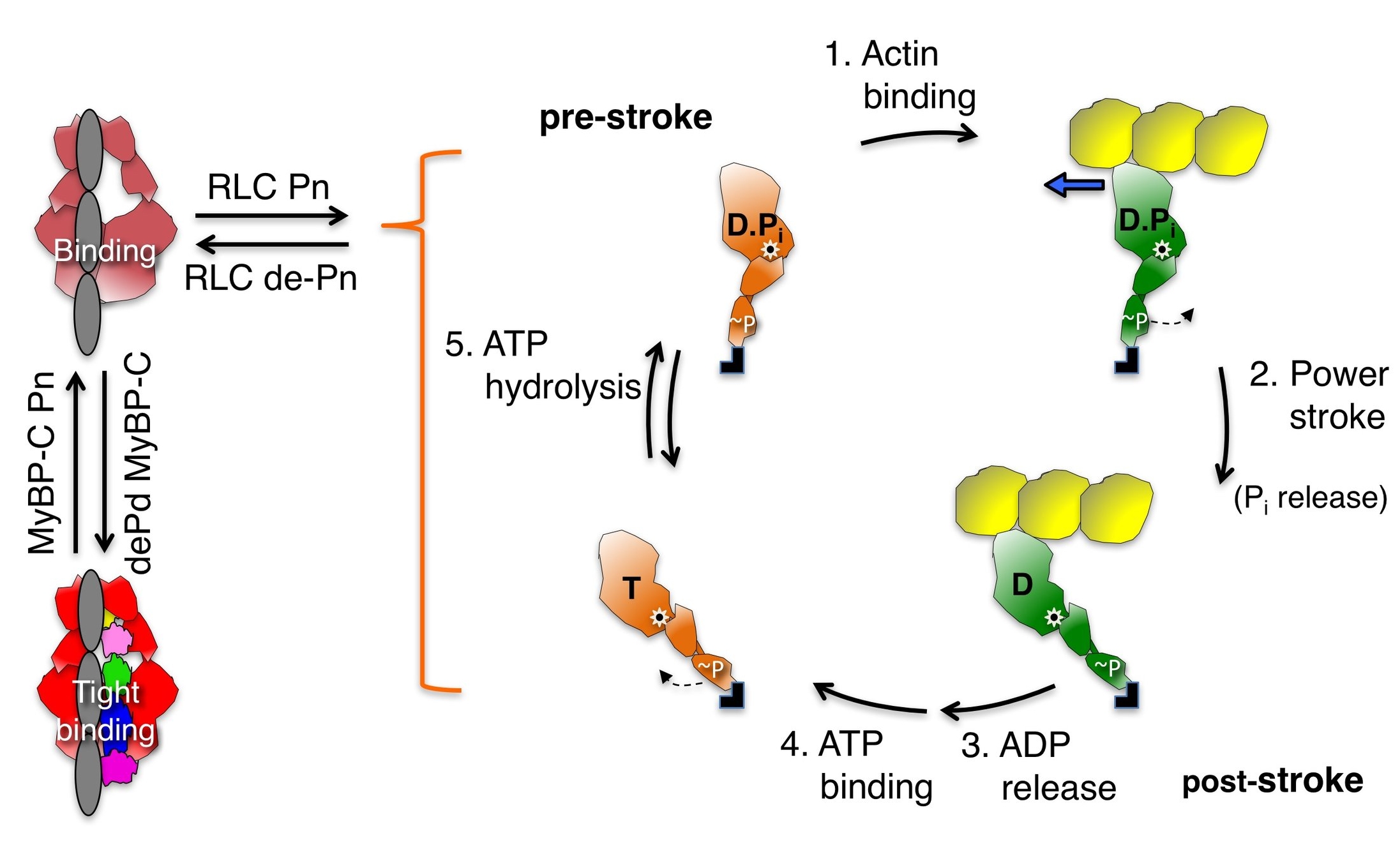
Regulation of the number of functionally accessible cardiac myosin heads for interaction with actin
3D homology models of full-length human beta-cardiac myosin S1 and HMM (shown as schematic structures in the figure above) can be downloaded from our DOWNLOADS site, and are described in detail there.
Our Research
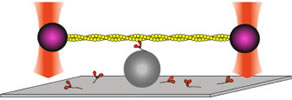
Our research interests have included the molecular basis of energy transduction that leads to ATP-driven myosin movement on actin, and the roles of the myosin family of molecular motors in eukaryotic cells. We have developed multiple new tools, including in vitro motility assays taken to the single molecule level using laser traps. We are now applying these tools toward an understanding of normal and diseased human cardiac function.